Your proven ally for clinical trials since 2002.
BiTrial is a Contract Research Organization (CRO) in the Central and Eastern European Region, providing clinical trial services for CRO’s, Pharma and Biotech companies, Medical Device and In-vitro Device manufacturers.
Our deep-rooted family values and patient-centric focus significantly accelerates drug and medical device development.
Achievements in 23 years:
Completed studies
0
Partner sites
0
Partner PI’s
0
Sponsors
0
Enrolled Patients
0
+
 Expertise
Expertise
Country Mix
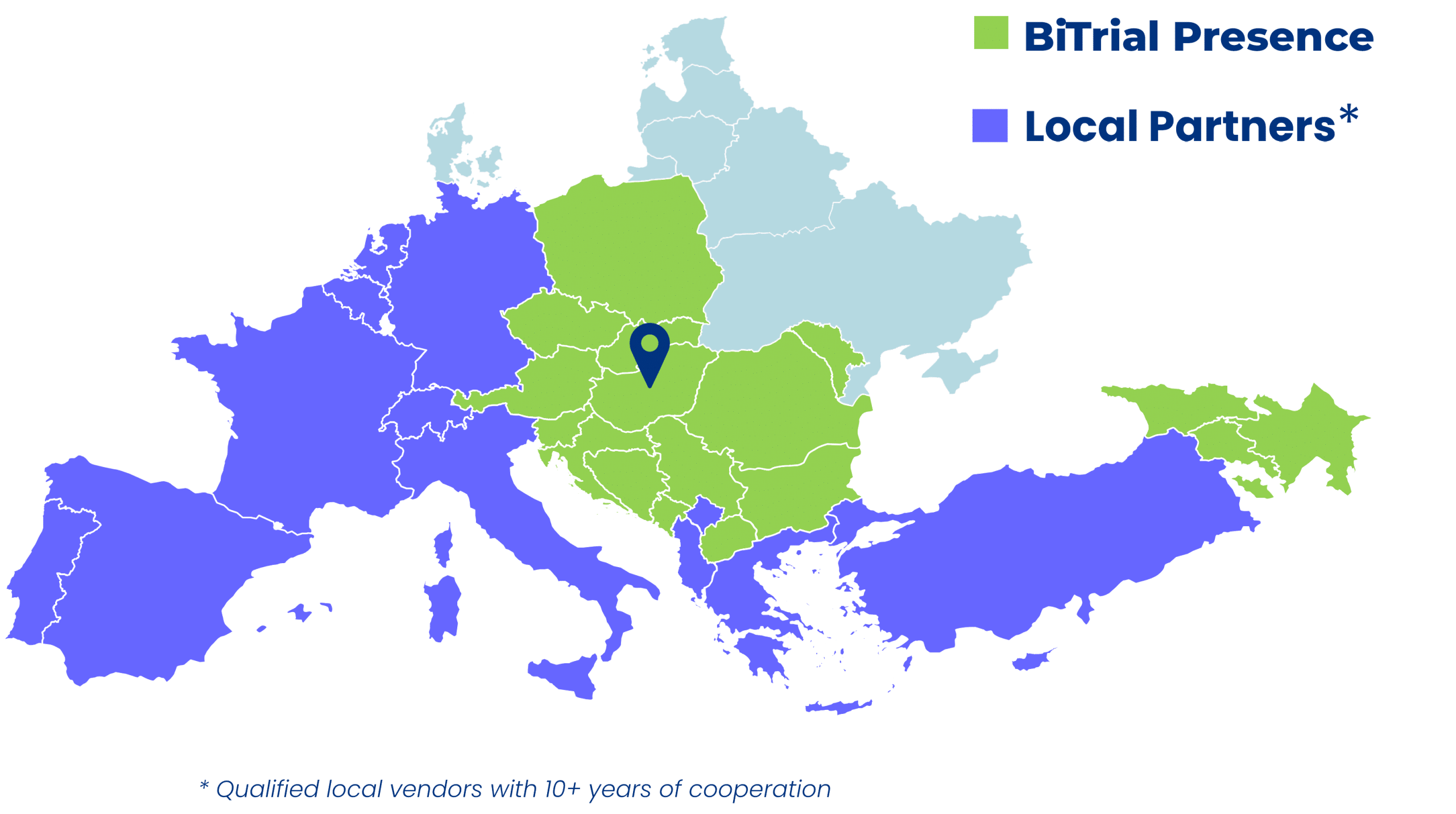
Armenia
Austria
Azerbaijan
Bosnia and Herzegovina
Bulgaria
Croatia
Czech Republic
Georgia
Hungary
Macedonia
Moldova
Poland
Romania
Serbia
Slovakia
Slovenia
Montenegro
Therapeutic Experience
![]() Partners
Partners










































































 Expertise
Expertise